 
Dated Tables of Atomic Weights published by the Commission refer to our best knowledge of the elements in natural terrestrial sources and have been published since 1902. To this end, on 30 March 1899 the invitation was sent to national scientific organizations worldwide to appoint delegates to the International Committee on Atomic Weights, which was formed in 1899, and consisted of 56 delegates from 11 countries. This committee published its first report in 1898, wherein it suggested the desirability of an International Committee on Atomic Weights. Landolt (Berlin University), serving as chair. The working committee consisted of Wilhelm Ostwald (University of Leipzig), Karl Seubert (University of Hanover), and Hans H. The creation of an international commission assigned with the evaluation of the atomic weights dates back to 1897, when soon-to-be Nobel laureate Hermann Emil Fischer proposed the creation of a working committee to report on atomic weights. Additionally, the Commission reviewed the recommendations to change the standard atomic weights of Yb (2015), Ar (2017), Ir (2017), Hf (2019), and Pb (2021) based on the review of published data on the isotopic composition of these elements, as well as the standard atomic weights of 14 other elements (2017, 2021) based on the Atomic Mass Evaluation 20 under the auspices of the International Union of Pure and Applied Physics (Al, Au, Co, F, Ho, Mn, Nb, Pa, Pr, Rh, Sc, Tb, Tm, Y). The recommendations included modifying the definition of normal materials and changing the expression of uncertainty of standard atomic weights and isotopic composition values. At these meetings, the Commission reviewed recommendations of its Subcommittees (Subcommittee on Natural Assessment of Fundamental Understanding of Isotopes, Subcommittee on Isotopic Abundance Measurements, and Subcommittee on Stable Isotope Reference Material Assessment). The Commission met in Tulln (Austria) from 3 to 4 August 2015, Groningen (The Netherlands) from 16 to 18 September 2017, in Berlin (Germany) from 3 to 4 July 2019 and in a virtual meeting 27 to 28 July 2021 under the chairmanship of Juris Meija. atomic mass " = color(green)(|bar(ul(color(white)(a/a)color(black)("47.The Commission on Isotopic Abundances and Atomic Weights (CIAAW, hereafter called the Commission) convenes biennially to evaluate recent developments in isotope measurement science and deliberate on related matters. The average atomic mass of titanium will thus be 
The decimal abundances for these five isotopes will be atomic mass" = sum_i m_"a i" xx "abundance of i"color(white)(a/a)|)))# 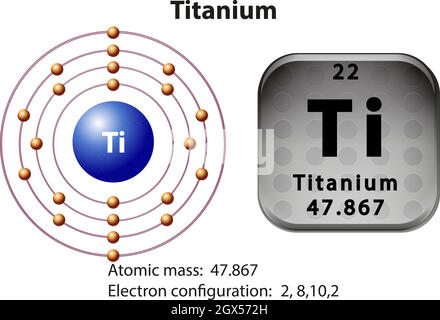
Simply put, each isotope #i# will contribute to the average atomic mass of the element in proportion to its decimal abundance, which is simply the percent abundance divided by #100#. Now, the average atomic mass of titanium is calculated by taking the weighted average of the atomic masses of its stable isotopes. When the problem doesn't provide you with the actual atomic mass of an isotope, #m_a#, you can use its mass number, #A#, as an approximation of its atomic mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
